.. By Sarah Rainsford
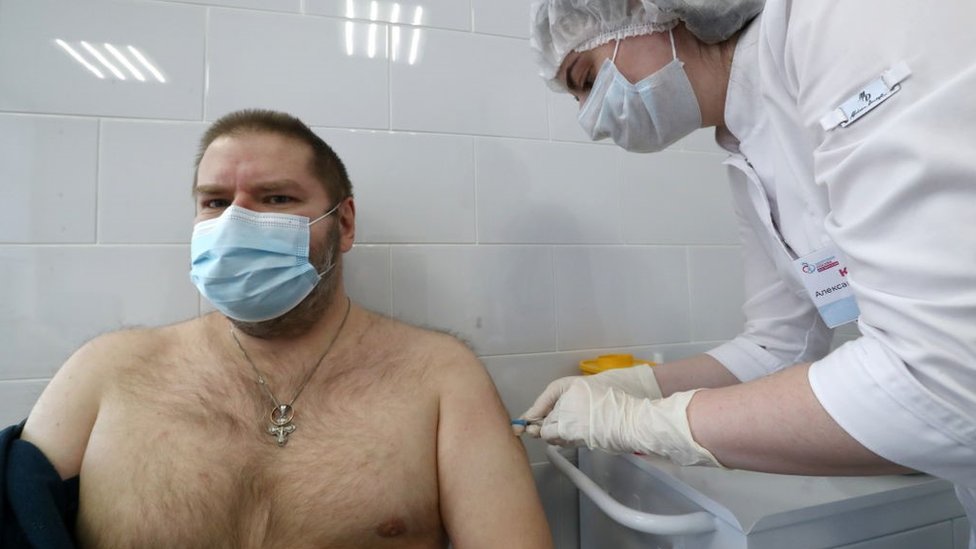 IMAGE COPYRIGHTGETTY IMAGES
IMAGE COPYRIGHTGETTY IMAGESIts name alone speaks of Russia’s ambition: Sputnik V, the country’s leading vaccine against Covid-19, is meant to be a world-beater just like its cosmic namesake.
Back in August, it was the first to be registered for emergency use although it had only been tested on a few dozen people.
Now doctors, teachers and social workers are being offered Sputnik V in a mass inoculation campaign ordered by President Vladimir Putin. Its timing, just ahead of a similar launch in the UK, is unlikely to be a coincidence.
But Sputnik V is still in the midst of trials to check that it’s safe and actually works, making some Russians wary of receiving it yet.
And despite a fanfare over the vaccine’s grand rollout, there are still problems scaling-up production.
The BBC’s Sarah Rainsford went to one central Moscow clinic where vaccines are taking place
In a sense, this is no “launch” at all. Russian health workers have been getting vaccinated in tandem with the official trials for several months. Teachers have been eligible too, and VIPs including President Putin’s own daughter have had the jab. The list is said to total more than 100,000 people.
Bold claims
“All of the staff here have been vaccinated,” nurse Oksana Konstantinova confirms, as she removes a tiny glass bottle of vaccine from the deep freeze at a Moscow clinic.
Sputnik V has to be stored at -18C at least in its liquid form. There are plans for a more practical, powder version of the vaccine but it’s not yet being made in large amounts.
Why did she accept an experimental jab? “We have to be able to say that the vaccine is safe and necessary in the current situation,” the nurse explains. “I was worried a bit. I am human! But I realised it was a fuss about nothing.”
Sputnik’s backers claim it offers 95% protection against the coronavirus, putting Russia’s offering right up alongside the vaccines of teams in the US and Europe. But here, the data released so far is based on interim results only – after just 39 trial volunteers caught Covid. That haste – coupled with such bold claims – has raised eyebrows.
“We hope the vaccine is effective, but it’s difficult to trust some of the figures,” argues Svetlana Zavidova, whose organisation monitors clinical trials in Russia.
She also lists concerns that Sputnik’s developers injected themselves with their own vaccine and that the product was registered for use after such limited trials.

“We don’t see the point of such a rush, other than announcing how we beat the rest of the world,” Zavidova says. “I think there’s a struggle between scientists and politicians, and the latter are winning.”
Limited issue
Like other countries, Russia presents its trial results to an external panel of experts for analysis. “I heard the interim reports and can say for sure that Sputnik is safe and can produce a short-term immune response,” oncologist Dr Alexander Rumyantsev told the BBC.
He had the vaccine himself in September and now boasts a large number of antibodies, though he said it was impossible to tell from the trial data how long any protection would last.
“We need more observation. I think we will only know that in spring.” Around 24% of volunteers experience side effects, the doctor said. “They were not pronounced, and they didn’t create any serious problems.”
But there have been problems for Sputnik of another kind. Its developers used to boast that tens of millions of doses of their vaccine would be rolling off conveyer belts this year, but they’ve had to scale-back expectations.
Even President Putin now expects a modest two million doses of Sputnik this month.
 IMAGE COPYRIGHTEPA
IMAGE COPYRIGHTEPAThe Russian vaccine comprises two entirely different injections – using different “vectors” or carriers for the coronavirus each time. Its backers insist that makes it more effective, but it also piles extra pressure on producers.
“Russia has taken on a crazy task,” Dmitry Morozov says, arguing that biotech firms like his should have been consulted on the practicalities at the very start.
“They basically doubled the work for us. It’s like you need to make two cars to actually move forward, but they’re both completely different: a Jeep and a mini bus,” he explains. “That’s a serious challenge. It means we have a far tougher task than most other manufacturers.”
Even so, Morozov is confident his firm, Biocad, can produce “millions” of doses of Sputnik. Its test batches are in Moscow now, waiting to be quality-cleared.

Another source told the BBC that producers have struggled with the second Sputnik jab in particular.
“The first component is the most stable, we can produce more of it,” the source said. “The second is less so and we have to keep strict temperature controls. I think it will soon be resolved.”
Tough sell
But even when production lines are up and running, there could be another issue to contend with.
We only saw a handful of people getting injected on the first day of “mass” vaccination. On Monday, a reporter for Novaya Gazeta described similar scenes at another Moscow clinic.
According to the paper, a nurse had to throw some Sputnik V away when she defrosted it and no-one turned up.
That’s a worry when the country is being hit hard by Covid, reporting close to 30,000 new cases a day. But it seems many Russians may have decided to wait.
“A lot of people don’t believe in the vaccine at this point,” Svetlana Zavidova believes. “I hope if there’s real scientific data, they will change their minds.”
.. Source